Former Opiate Manufacturer CEO Arrested in ArizonaBillionaire Jonathan Kapoor is charged with violating antikickback laws
On Oct. 26, 2017, the former CEO of the opiate manufacturer Insys Therapeutics, Inc. Jonathan Kapoor, was arrested on fraud and racketeering charges, NPR reports. The arrest was made in […]
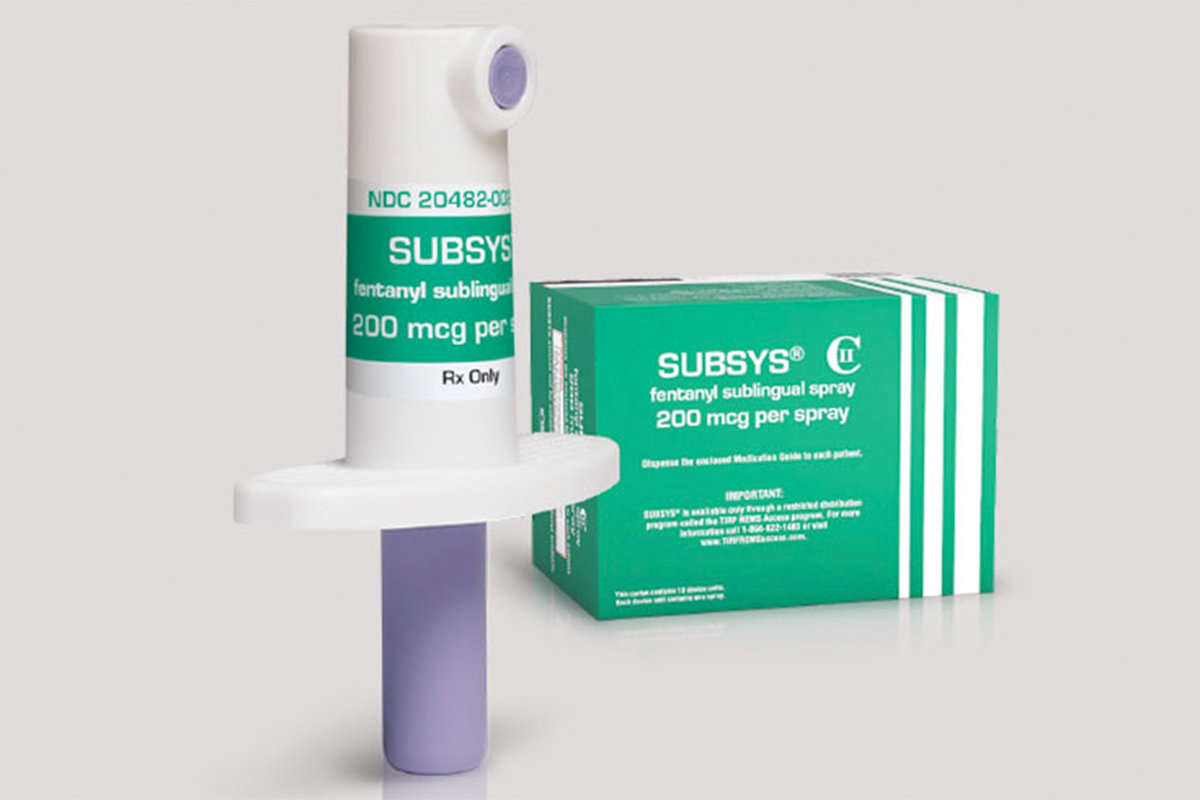
On Oct. 26, 2017, the former CEO of the opiate manufacturer Insys Therapeutics, Inc. Jonathan Kapoor, was arrested on fraud and racketeering charges, NPR reports. The arrest was made in connection with a kickback scheme in which company representatives paid healthcare providers to prescribe a potent painkiller meant only for cancer patients for “off-label” use. The painkiller, Subsys, is a spray form of the drug fentanyl, a short-acting opiate that is 50 to 100 times more powerful than morphine.
According to the Insys Therapeutics website, Subsys is specifically designed to treat breakthrough cancer pain in patients who are already taking a long-acting opiate. Breakthrough cancer pain occurs when a person with stable, persistent pain experiences a sudden increase in pain intensity. These episodes are usually brief, lasting anywhere from a minute to several hours. Nevertheless, they can be very debilitating and make overall pain management extremely challenging.
Unlike oral opiates, which take time to be absorbed from the gastrointestinal tract, Subsys is sprayed under the tongue. Because this area contains dozens of tiny blood vessels, the medicine is absorbed into the bloodstream very quickly. It provides a significant degree of pain relief within 15 minutes, with peak effect after one hour, according to Insys. This makes it an excellent medicine for people with breakthrough cancer pain.
However, it also means the drug is very dangerous and has an enormous potential for abuse.
Illegal Kickbacks
According to prosecutors, Kapoor and other high-level executives at Insys Therapeutics engaged in a nationwide conspiracy to bribe healthcare practitioners to write large numbers of prescriptions for Subsys. Many of the doctors who wrote the prescriptions owned pain clinics. And most of the patients who received the prescriptions did not have cancer, the indictment claims.

Credit: forbes.com
Prosecutors have also charged Kapoor and other co-defendants with insurance fraud for deceiving insurance companies in an effort to get them to approve payment for the drug. The company did so by organizing a “reimbursement unit,” the purpose of which was solely to encourage insurance companies and benefits managers to approve the drug for off-label use.
Both the District Attorney’s office and federal law enforcement condemned the scheme, calling Insys executives “no better than street level drug dealers.” In a press release from the United States Attorney’s Office in the District of Massachusetts, which issued the indictment, Acting U.S. Attorney William D. Weinreb commented:
“In the midst of a nationwide opioid epidemic that has reached crisis proportions, Mr. Kapoor and his company stand accused of bribing doctors to overprescribe a potent opioid and committing fraud on insurance companies solely for profit…..We must hold the industry and its leadership accountable – just as we would the cartels or a street-level drug dealer.”
No charges have yet been filed against the healthcare practitioners involved in the scheme. However, according to a 2016 Forbes report, law enforcement officials in Michigan, Florida, Kansas, New Hampshire, Rhode Island and New York are investigating doctors who worked with Insys.












Leave a Reply